In October of 2016, Theralase, a small Laser Therapy company located in Toronto, distributed a questionnaire to BioFlex Laser Medicine Practitioners. Several recipients were confused with regard to the origin of this document. This is understandable as it is a thinly disguised effort to make it appear to have originated at Meditech. I can assure you that this was not the case and unfortunately, this demonstrates the characteristics of some industry principals – projecting a blend of hucksterism, hype, disinformation and deception.
Most significantly, the questionnaire reflects the lack of clinical understanding that prevails within some Laser Industry Manufacturers, as reflected by their literature, press releases, etc., best described as pseudoscience. These communications perform a disservice to the legitimate sector of Laser Medicine, aside from the paucity of comprehension of clinical pathologies and the inappropriate design of devices required to resolve the latter. All manufacturers must focus on the design and building of machines to relieve the many problems that exist, instead of simply building devices to generate profits. Without fully understanding these realities and objectives, their efforts will continue to be self-defeating.
Prior to responding to the five questions included in the questionnaire, we will attempt to provide some overall perspective to this entire situation. Moreover, we regard this as an opportunity to expand the educational process that is so important in qualifying and empowering Laser Therapists.
At this time, at least three knowledgeable and respected medical practitioners, offended by the Theralase comments, have offered to respond to the questionnaire and these reports will be delivered to you sometime in April.
At Meditech, we are reluctant to advertise however we make efforts to share information reflecting current basic research and engineering improvements, many of which are integrated into the systems we manufacture, along with new, effective clinical applications. We prefer to leave the evaluation of our technology and the clinical results achieved to others who have the ability to independently comment on our body of work and how it is affecting current medical practice. To wit:
- Dr. Norman Doidge’s book, “The Brain’s Way of Healing”, which was published in 2015, devotes an entire chapter to BioFlex Laser Technology and this text still resonates with the medical and lay community in many parts of the globe.
- In 2015, an Australian TV network filmed our activities over the course of two weeks and developed a documentary, extoling our technology in Australia, New Zealand, Tasmania and adjacent regions. The network called Seven Network Limited is similar to the U.S. program, 60 Minutes.
- In October 2016, Meditech was featured on David Suzuki’s “The Nature of Things”, which will be broadcast in many countries around the world over the next several years. This achieved considerable favourable international recognition and I stress that this exposure was not solicited.
- In December 2016, Dr. Gifford-Jones, a highly respected syndicated medical journalist wrote an article which was circulated in over 70 newspapers in Canada. The article commented favourably on our treatment of cerebral concussion and other neurological conditions.
- In addition, many articles have been published with regard to Meditech’s Laser Technology based on our achievements in this field in a number of countries.
At this time, I enclose a prescient article written independently by Dr. Leonard Rudnick, the director of Laser Therapeutics in Tuscon, Arizona. This commentary was written in July 2007 in response to previous comments by Theralase that we originally chose to ignore as being nonsensical, much like the current five questions recently circulated. This article was written over ten years ago and is evidence of the negative trend that Theralase is intent on pursuing.






I also include an article presented by Dr. Jan Tuner, who is based in Sweden and has been highly active in the Laser Therapy sector for over 30 years. Dr. Tuner’s comments with regard to the minimal value on tissue healing of light delivered at 905 nM, the wavelength recommended by Theralase, is particularly relevant. A number of other issues are also addressed in this communication.





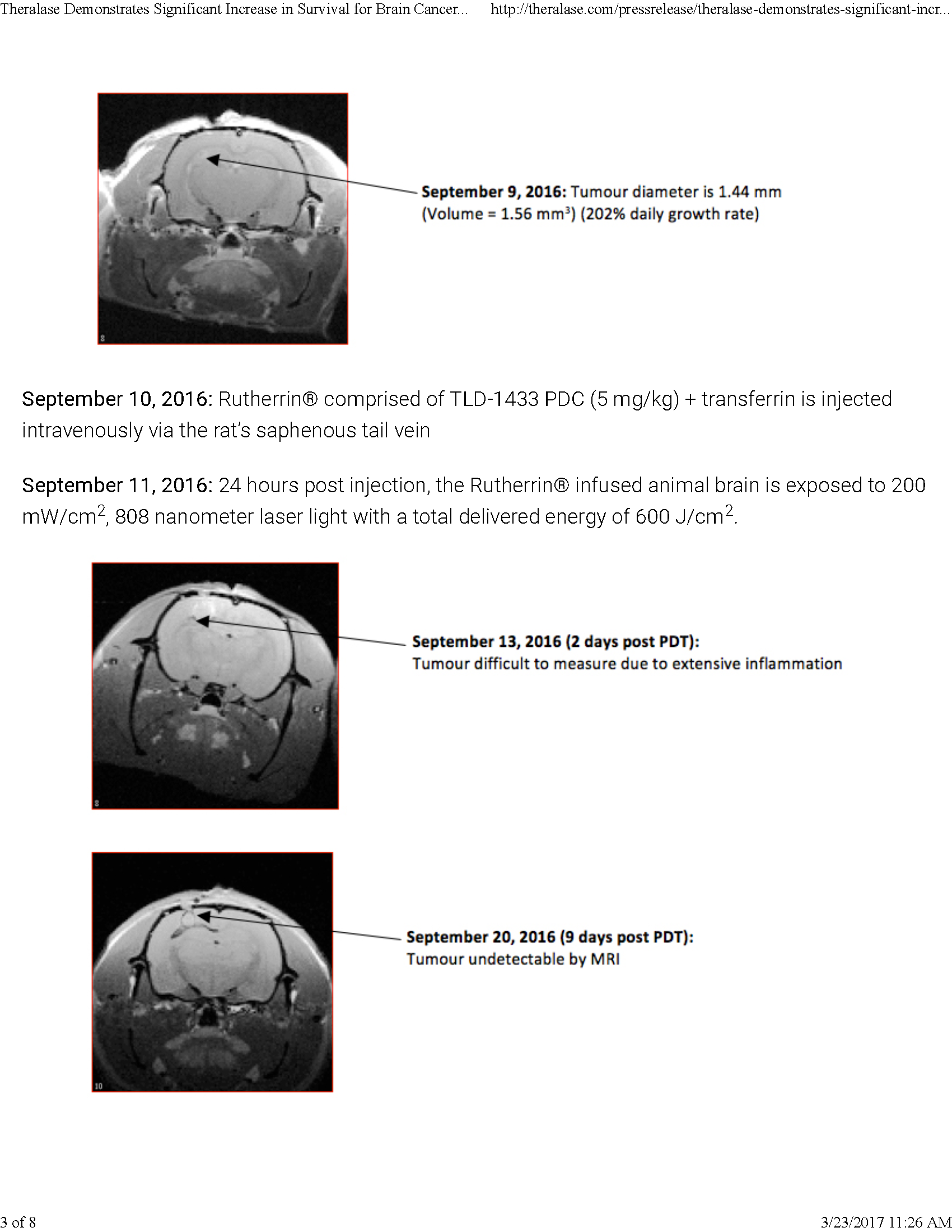
On a final note, I refer to a typical Theralase press release sent to me by a biotechnology analyst for my comment. The release is dated October 13, 2016. After reviewing the document, my conclusion is that a company that would release a paper of this nature has a limited understanding of research, science and integrity. Performing a study of brain cancer on a single rate, regardless of the outcome, is irrelevant at best.
Unfortunately in our society, there are no controls over this type of deceptive literature. Our conclusion is that prevarication should not be the standard for success and hopefully over time, activities of this nature should lead to the demise of the company.
Another penny stock disappearing from the Venture Exchange will certainly not be missed.






Conclusion
Some good emanates from all events, no matter what the intentions may be. This matter has provided an opportunity for Meditech to expand the educational process and clarify a number of contentious issues. The answers to the questionnaire that Theralase has posed and answered inappropriately will be fully resolved by a follow-up communication, which you should receive in April 2017.
Fred Kahn MD, FRCS(C)
CEO, Meditech International Inc.